Virtual Contrast Supervision IT Security: HIPAA, HITECH and Vendor Essentials


Key Takeaways
- HIPAA and HITECH compliance is not optional for virtual contrast supervision platforms. Both laws impose specific technical, administrative, and physical safeguards that directly govern how patient data is handled during remote sessions.
- End-to-end encryption, multi-factor authentication, and audit logging are the three non-negotiable technical controls every virtual supervision vendor must demonstrate before you sign a Business Associate Agreement.
- HITECH significantly increases HIPAA penalties, especially for willful neglect, meaning a single unsecured video session could trigger fines in the millions.
- Vendor vetting is a compliance requirement, not a best practice; imaging centers are legally accountable for the security posture of every third-party platform that touches Protected Health Information (PHI).
- At ContrastConnect, we provide CMS-compliant remote contrast coverage, supervising more than 75,000 exams each month with response times measured in seconds and a documented zero missed-response record across 130+ contrast reactions managed monthly, all within HIPAA and HITECH compliance.
Virtual Contrast Supervision Creates Real Security Obligations
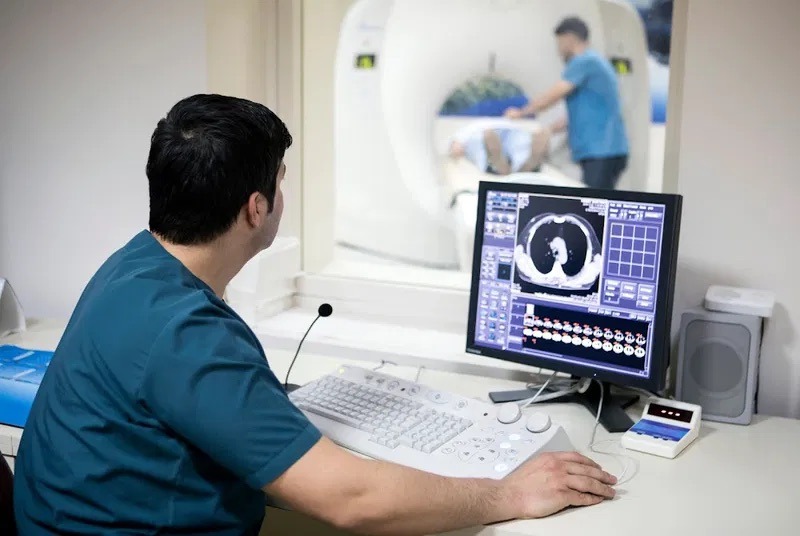
Most imaging centers focus on clinical workflows when adopting virtual contrast supervision, but the IT security obligations underlying that decision are where compliance risk actually lies.
The moment a radiologist views a patient's imaging session remotely, PHI is in motion. Real-time video feeds, patient identifiers, diagnostic communications, and session metadata all qualify as PHI under HIPAA's definition. This means every component of your virtual supervision stack is a potential compliance liability if it isn't properly secured.
Unlike standard telehealth platforms, virtual contrast supervision operates in a high-acuity environment where technical failures are patient safety events. The security architecture has to account for both. That dual pressure is what makes this platform category uniquely demanding from an IT security standpoint.
This article discusses key aspects of virtual contrast supervision, including IT security and the relevant HIPAA and HITECH policies.
What HIPAA Actually Requires for Virtual Supervision Systems
The Three Safeguard Categories That Apply Directly
HIPAA's Security Rule lays out the ground rules. It applies to all electronic PHI (ePHI), which is exactly what flows through a virtual supervision session. The rule doesn't prescribe specific technologies, but it does mandate that covered entities and their business associates implement safeguards across three distinct categories: administrative, physical, and technical.
Breaking this down practically for virtual contrast supervision environments, here is how each safeguard category maps to real system components:
- Administrative: Risk assessments covering the virtual platform, workforce training on secure session protocols, and vendor management policies, including signed Business Associate Agreements (BAAs).
- Physical: Workstations use policies for remote radiologists, screen privacy controls, and secure device management, including auto-logoff settings.
- Technical: End-to-end encryption for all session data, role-based access controls, unique user authentication, automatic session timeouts, and full audit trail logging.
End-to-End Encryption Is Non-Negotiable
Any virtual supervision platform transmitting ePHI must encrypt that data both in transit and at rest. The accepted standard for transmission encryption is TLS 1.2 or higher, with TLS 1.3 now considered best practice. For stored session data, including recordings, logs, and metadata, AES-256 encryption is the healthcare industry benchmark. If a vendor cannot confirm both standards in writing, that is an immediate disqualifier.
Audit Logs & Access Controls for Remote Sessions
HIPAA requires that every access to ePHI be logged and that those logs be reviewable. For virtual supervision sessions, this means your platform must record who accessed the session, when they accessed it, from which device and IP address, and what actions were taken. These logs must be tamper-evident, retained for a minimum of six years, and accessible for compliance audits.
Multi-Factor Authentication Requirements
Multi-factor authentication (MFA) is classified as an addressable implementation specification under HIPAA. It means you must either implement it or document a specific, justified alternative that provides equivalent protection. In practice, any virtual supervision platform that doesn't support MFA should be considered non-compliant for healthcare use.
How HITECH Raises the Stakes Beyond HIPAA
The Health Information Technology for Economic and Clinical Health (HITECH) Act, enacted in 2009, dramatically expanded both the reach and the teeth of HIPAA enforcement. Where HIPAA established the rules, HITECH established the consequences.
HITECH introduced a tiered civil penalty structure, extended HIPAA obligations directly to business associates, and created mandatory breach notification requirements that apply whether the breach was intentional or not.
Breach Notification Rules That Affect Virtual Platforms
Under HITECH's Breach Notification Rule, any unauthorized access to unsecured ePHI triggers mandatory notification obligations. For a virtual supervision platform, a breach could be as straightforward as an unencrypted session recording stored on an unsecured server, or as complex as a man-in-the-middle attack on an inadequately protected video stream.
The notification timeline is strict: affected individuals must be notified within 60 days of breach discovery, and breaches affecting 500 or more individuals require HHS notification without unreasonable delay and no later than 60 days after discovery. If 500 or more residents of a single state or jurisdiction are affected, prominent media notification in that area is also required.
Increased Penalties for Willful Neglect
HITECH created four penalty tiers based on culpability, and the numbers are serious. Willful neglect that is not corrected carries a minimum penalty of $50,000 per violation under HITECH's original penalty structure, with an annual maximum of $1.5 million for identical violations under OCR's current enforcement discretion.
A single unsecured virtual supervision session that exposes PHI where the covered entity knew about the security gap and failed to fix it can land in that top tier. For imaging centers operating on thin margins, that kind of exposure isn't theoretical. It's existential.
Vendor Management Is a Compliance Requirement, Not a Suggestion

Under both HIPAA and HITECH, imaging centers are directly accountable for the security practices of every vendor that touches their ePHI. That accountability doesn't transfer when you sign a contract; it expands. You're now responsible for your own compliance and for ensuring your vendor maintains equivalent standards.
What a Business Associate Agreement Must Cover
A Business Associate Agreement (BAA) is the legal contract that binds your virtual supervision vendor to HIPAA's security and privacy standards. Without a signed BAA, any ePHI shared with that vendor is an unauthorized disclosure.
A compliant and protective BAA for a virtual supervision platform must explicitly address the following elements:
- The specific types of ePHI the vendor will access, store, or transmit.
- Permitted uses and disclosures of that ePHI and strict prohibitions on all others.
- The vendor's obligation to implement HIPAA-required safeguards and report breaches within the HITECH-mandated 60-day window.
- Sub-contractor management requirements confirming the vendor's downstream partners are also bound by equivalent BAAs.
- Data return or destruction requirements at contract termination.
- The covered entity's right to audit the vendor's compliance posture.
How to Vet a Virtual Supervision Vendor's Security Posture
Vetting a vendor's security posture goes well beyond asking if they're "HIPAA compliant." Any vendor can claim compliance, so what you need is documented, verifiable evidence. Start by requesting their most recent SOC 2 Type II audit report.
This report, produced by an independent third-party auditor, evaluates a vendor's security controls across availability, confidentiality, processing integrity, and privacy over a sustained audit period, typically six to twelve months. A SOC 2 Type I report only reflects a single point in time and provides significantly less assurance.
Get CMS-Compliant Remote Contrast Supervision with ContrastConnect

When it comes to protecting patient data and meeting the stringent security demands of virtual contrast supervision, choosing the right partner matters as much as choosing the right technology. At ContrastConnect, our radiologist-owned platform was built from the ground up to meet HIPAA, HITECH, and CMS compliance requirements.
Every component of our infrastructure, from encrypted telehealth connections to audit-ready documentation and discharge summaries, is designed to satisfy the necessary regulatory standards. Our qualified radiologists supervise more than 75,000 contrast exams monthly across outpatient imaging facilities and hospital networks nationwide, managing over 130 contrast reactions each month, with a documented zero-missed-response safety record.
If your organization is preparing for CMS's anticipated permanent adoption of virtual supervision in 2026, now is the time to ensure your vendor infrastructure meets every HIPAA, HITECH, and CMS benchmark covered in this guide. We give you a cost-efficient alternative to onsite staffing that lets you reduce cancellations, extend operating hours, and scale imaging operations, all without compromising the security and compliance standards your patients and regulators expect.
Start your coverage assessment today.
Frequently Asked Questions (FAQs)
What encryption standard is required for HIPAA-compliant virtual contrast supervision platforms?
HIPAA-compliant virtual supervision platforms must use TLS 1.2 or higher for data in transit, with TLS 1.3 now considered best practice. For data at rest, including session logs, recordings, and metadata, AES-256 is the accepted healthcare industry standard. Any vendor that cannot confirm both encryption standards in writing should not be granted access to your ePHI.
Does HITECH apply to imaging centers using third-party virtual supervision vendors?
Yes. HITECH extended HIPAA obligations to business associates, meaning both the imaging center and the virtual supervision vendor are independently accountable for compliance. The imaging center remains liable if it fails to conduct adequate vendor due diligence or neglects to execute a compliant BAA.
Do technologists need certification before using remote supervision platforms?
There is no single federal certification requirement for technologists using virtual supervision platforms, but HIPAA's administrative safeguard requirements mandate workforce training on security policies and procedures. This means every staff member who interacts with the virtual supervision platform must receive documented training on secure session protocols, PHI handling, and incident reporting procedures before they are permitted to use the system.
What happens if a virtual supervision platform experiences a data breach during a session?
The session must be terminated, and the affected system must be isolated from the network to prevent further exposure. Next, the virtual supervision vendor must be notified immediately, and your organization's documented incident response plan must be activated within hours of discovery, not days.
Breaches affecting 500 or more individuals require notification to the HHS Office for Civil Rights without unreasonable delay and no later than 60 days after discovery. If the breach affects 500 or more residents of a single state or jurisdiction, a prominent media notice in that area is also required under HITECH.
How is ContrastConnect more reliable than other remote contrast supervision providers?
ContrastConnect is radiologist-owned and purpose-built for contrast supervision, with qualified radiologists overseeing more than 75,000 exams monthly and treating over 130 contrast reactions each month.
Our services are backed by a documented zero-missed-response safety record, and our always-on platform delivers response times measured in seconds, with audit-ready documentation.
*Note: Information provided is for general guidance only and does not constitute medical, legal, or financial advice. Pricing estimates and regulatory requirements are current at the time of writing and subject to change. For personalized consultation on imaging center operations and virtual contrast supervision, contact ContrastConnect.
Trusted Nationwide





































.avif)











.avif)











.avif)




1,000,000
Contrast exams supervised annually
75,000+
Hours of supervision monthly
3,900+
Technologists certified
100s
Of imaging partners nationwide
130+
Contrast reactions treated monthly
100%
Requested hours covered
Connect with us.


